ST. PAUL, Minn.--(BUSINESS WIRE)--
Adhesives play a critical role in the success of a medical device, whether it’s holding the device together or adhering it to the wearer’s skin. But with so many adhesive products available, it can be a challenge for design engineers to know which one is right for their project. To help bridge this critical gap, 3M developed a “Find My Adhesive” product selector tool to help make medical adhesive selection a simpler process. The product selector tool will be available to demo at CES 2018 in Las Vegas from January 9-12, 2018 in the 3M booth located in Sands, Halls A-D; 43517 Health & Wellness Marketplace.
This press release features multimedia. View the full release here: http://www.businesswire.com/news/home/20180103005235/en/
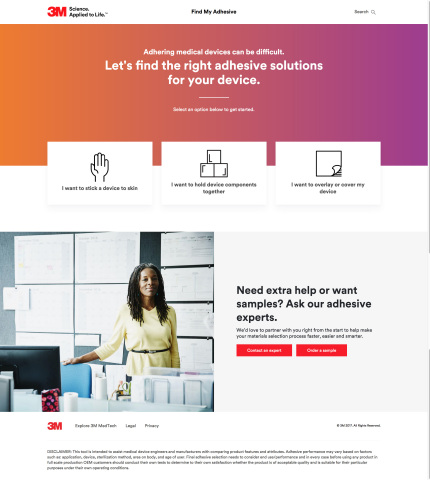
3M's FindMyAdhesive.com is an interactive online tool designed to make medical adhesive selection simple for engineers by asking a series of project-specific questions. (Photo: 3M)
“Adhering medical devices can be a difficult task. Using the wrong adhesive could result in unforeseen added costs and device development delays,” said Marcello Napol, global business director in 3M’s Critical and Chronic Care Solutions Division. “Our goal with Find My Adhesive is to leverage our more than 50 years of adhesive research and development expertise to provide a useful tool to design engineers that helps streamline the adhesive selection process and improve the device’s overall probability of success. The interactive, intuitive and efficient tool can assist design engineers in an oftentimes unexpectedly challenging and critical part of the medical device design process.”
Find My Adhesive is an online resource that uses a series of project-specific questions to identify a list of the most appropriate medical adhesive suggestions. Each question is centered on an important consideration, such as the adhesive’s purpose (stick-to-skin, hold device components together or overlay / cover my device) and needs, such as wear time, the material you need to stick to and intended user, which works to narrow down the adhesives recommended for use. It’s free to use and, unlike other selection tools, does not require an account to access. If projects require extra assistance and feedback, users can contact a 3M medical adhesives expert directly through the site.
To help narrow down the right adhesive for your next medical device project, the Find My Adhesive product selector tool can be found at findmyadhesive.com, or check it out at CES in the 3M booth during show hours.
To learn more about overcoming design challenges of adhesives, attend 3M medical adhesives expert Dr. Diana Eitzman’s panel session titled “Biometrics boost their numbers by losing friction,” where she’ll speak about the science of skin, including skin dynamics that impact wearable medical device design. This Digital Health Summit panel will be on Wednesday, January 10 from 4:40-5:00 p.m. at the Venetian, Level 4, Lando 4304.
3M is a trademark of 3M Company.
About 3M
At 3M, we apply science in collaborative ways to
improve lives daily. With $30 billion in sales, our 90,000 employees
connect with customers all around the world. Learn more about 3M’s
creative solutions to the world’s problems at http://www.3m.com/
or on Twitter @3M or @3MNewsroom.
View source version on businesswire.com: http://www.businesswire.com/news/home/20180103005235/en/




